3-Acetamidophthalic anhydride
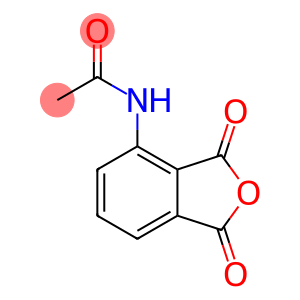
N-(1,3-Dioxo-1,3-dihydro-2-benzofuran-4-yl)acetamide
CAS: 6296-53-3
Molecular Formula: C10H7NO4
3-Acetamidophthalic anhydride - Names and Identifiers
| Name | N-(1,3-Dioxo-1,3-dihydro-2-benzofuran-4-yl)acetamide |
| Synonyms | NSC 16261 3-Acetamidophthalic anhydride 3-AcetaMidophthalic anhydride 3-AcetylaMinophthalic Anhydride 3-AcetylaMino-phthalsaeure-anhydrid N-(1,3-Dioxo-1,3-dihydro-2-benzofuran-4-yl)acetaMide N-(1,3-Dihydro-1,3-dioxoisobenzofuran-4-yl)acetamide N-(1,3-Dioxo-1,3-dihydro-2-benzofuran-4-yl)acetamide AcetaMide, N-(1,3-dihydro-1,3-dioxo-4-isobenzofuranyl)- acetamide, N-(1,3-dihydro-1,3-dioxo-4-isobenzofuranyl)- |
| CAS | 6296-53-3 |
| EINECS | 808-040-8 |
| InChI | InChI=1/C10H7NO4/c1-5(12)11-7-4-2-3-6-8(7)10(14)15-9(6)13/h2-4H,1H3,(H,11,12) |
3-Acetamidophthalic anhydride - Physico-chemical Properties
| Molecular Formula | C10H7NO4 |
| Molar Mass | 205.17 |
| Density | 1.512±0.06 g/cm3(Predicted) |
| Melting Point | 185-186℃ |
| Boling Point | 489.1±28.0 °C(Predicted) |
| Flash Point | 249.6°C |
| Solubility | Chloroform (Slightly, Sonicated), Ethyl Acetate (Slightly, Sonicated), Methanol |
| Vapor Presure | 1.03E-09mmHg at 25°C |
| Appearance | Solid |
| Color | Off-White to Pale Yellow |
| pKa | 13.81±0.20(Predicted) |
| Storage Condition | Inert atmosphere,Room Temperature |
| Stability | Moisture Sensitive |
| Refractive Index | 1.657 |
| MDL | A151652 |
3-Acetamidophthalic anhydride - Reference Information
| Application | 3-acetylaminophthalic anhydride is mainly used for the synthesis of APREMILAST. In addition, it may also modulate intracellular pro-inflammatory and anti-inflammatory cytokine effects by increasing intracellular c AMP levels by being specific for cyclic adenosine monophosphate (c AMP). |
| preparation | preparation of rhodium (I) BIS (1, 5-cyclooctadiene) trifluoromethanesulfonate under nitrogen A mixture of (9mg,0.018 mmol), 2,2, 2-trifluoroethanol (3 mL) and the appropriate ligand (0.037 mmol) or pre-formed complex of ligand with metal (0.018 mmol). Then, 1-(3-ethoxy-4-methoxyphenyl)-2-(methylsulfonyl) ethylamine (0.37 mg, mmol) was added. The resulting mixture was heated to 50 °c. And hydrogenated at 250psig hydrogen pressure. After 18 hours, the mixture was removed from the hydrogenator and passed through achiral HPLC(Hypersil BDS C8,5.0 pm,250x 4.6mm,1.5/min,278 nm, A gradient of 90/10 to 80/20 0.1% TFA/MeOH in water followed by a gradient to 10/90 0.1% TFA/MeOH in water over the next 15 minutes) was monitored. Also by chiral HPLC(Chiralpak AD-H 5.0 pm Daicel,250x 4.6 mm,1.0 mL/min,280 nm,70:30:0.1 heptane-1-PrOH-diethylamine or 50:40:10:0.0 heptane-EtOH-i-PrOH-diethylamine) the chiral purity of reactions containing> 10 Area% product enamine was determined, further purification gave the APREMILAST intermediate. |
Last Update:2024-04-09 21:04:16
Supplier List
Spot supply
Product Name: N-(1,3-Dioxo-1,3-dihydroisobenzofuran-4-yl)acetamide Visit Supplier Webpage Request for quotationCAS: 6296-53-3
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: N-(1,3-Dioxo-1,3-dihydro-2-benzofuran-4-yl)acetamide Request for quotation
CAS: 6296-53-3
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
CAS: 6296-53-3
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
Spot supply
Product Name: 3-Acetamidophthalic anhydride Visit Supplier Webpage Request for quotationCAS: 6296-53-3
Tel:
Email: qianyanbiochem@gmail.com
Mobile: 13247110337
QQ: 2972965813
Product List: View Catalog
Product Name: 1,3-Dioxo-2-isoindolineaceticacid Request for quotation
CAS: 6296-53-3
Tel: +86 17531972939
Email: anna@api-made.com
QQ: 905983447
WhatsApp: +86 17531972939
CAS: 6296-53-3
Tel: +86 17531972939
Email: anna@api-made.com
QQ: 905983447
WhatsApp: +86 17531972939
Product Name: 3-Acetamidophthalic anhydride Visit Supplier Webpage Request for quotation
CAS: 6296-53-3
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 6296-53-3
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Spot supply
Product Name: N-(1,3-Dioxo-1,3-dihydroisobenzofuran-4-yl)acetamide Visit Supplier Webpage Request for quotationCAS: 6296-53-3
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: N-(1,3-Dioxo-1,3-dihydro-2-benzofuran-4-yl)acetamide Request for quotation
CAS: 6296-53-3
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
CAS: 6296-53-3
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
Spot supply
Product Name: 3-Acetamidophthalic anhydride Visit Supplier Webpage Request for quotationCAS: 6296-53-3
Tel:
Email: qianyanbiochem@gmail.com
Mobile: 13247110337
QQ: 2972965813
Product List: View Catalog
Product Name: 1,3-Dioxo-2-isoindolineaceticacid Request for quotation
CAS: 6296-53-3
Tel: +86 17531972939
Email: anna@api-made.com
QQ: 905983447
WhatsApp: +86 17531972939
CAS: 6296-53-3
Tel: +86 17531972939
Email: anna@api-made.com
QQ: 905983447
WhatsApp: +86 17531972939
Product Name: 3-Acetamidophthalic anhydride Visit Supplier Webpage Request for quotation
CAS: 6296-53-3
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 6296-53-3
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
View History




